Biopharmaceutical companies, also known as biotech or biopharma companies, are organizations that develop, manufacture, and market drugs or therapies based on biological compounds. These companies focus on research and development (R&D) of new drugs and therapies using biotechnology processes, such as genetic engineering, recombinant DNA technology, and cell therapy.
Biopharmaceutical companies typically follow a series of phases or stages in their operations, which can be broadly categorized as follows.

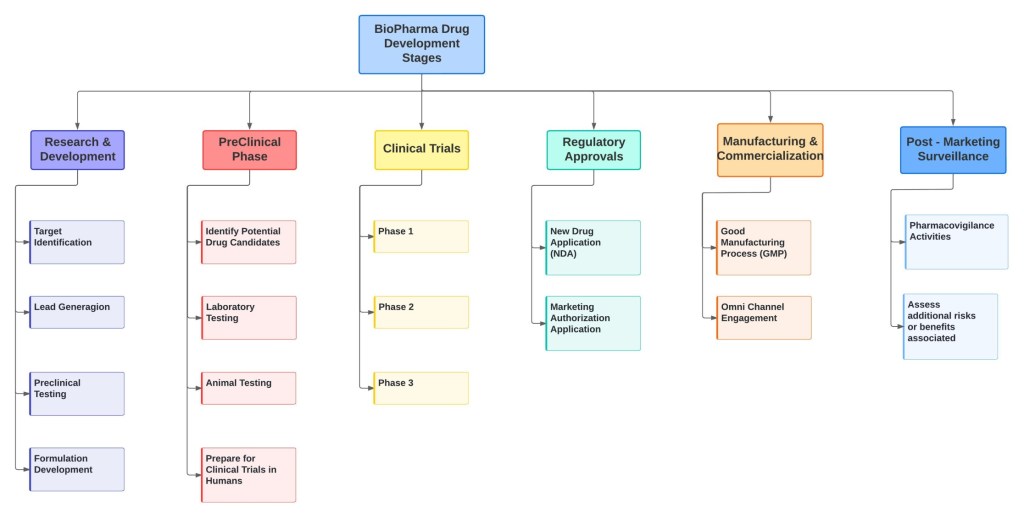
1. Research and Development (R&D) Phase: This phase involves extensive research and development activities to identify potential drug candidates or therapeutic approaches. It includes target identification, lead generation, preclinical testing, and formulation development. This phase is focused on understanding the biological mechanisms underlying diseases and exploring ways to develop effective treatments.
2. Preclinical Phase: In the preclinical phase, the potential drug candidates undergo rigorous testing in laboratories and animal models to assess their safety, efficacy, and toxicity profiles. This phase helps gather essential data to support the subsequent clinical trials in humans.
3. Clinical Trials: Clinical trials are conducted to evaluate the safety, efficacy, and dosage requirements of the drug candidate in human subjects. Clinical trials typically consist of three main phases:
Phase 1: In this initial phase, a small group of healthy volunteers or patients receive the investigational drug. The primary focus is on evaluating safety, dosage, and potential side effects.
Phase 2: This phase involves a larger group of patients and further investigates the drug’s effectiveness and optimal dosage. It also provides additional safety data.
Phase 3: Phase 3 trials involve a larger number of patients and are designed to confirm the drug’s effectiveness, monitor side effects, and compare it with existing treatments or placebos. These trials provide the data necessary for regulatory approval.
4. Regulatory Approval: Once the clinical trials are completed, the biopharmaceutical company submits a New Drug Application (NDA) or a Marketing Authorization Application (MAA) to regulatory authorities such as the U.S. Food and Drug Administration (FDA) or the European Medicines Agency (EMA). The regulatory agencies review the data to assess the drug’s safety, efficacy, and quality before granting approval.
5. Manufacturing and Commercialization: If the regulatory authorities approve the drug, the company proceeds with large-scale manufacturing, ensuring quality control and adherence to Good Manufacturing Practices (GMP). Once the drug is manufactured, it is distributed and made available to healthcare providers, patients, and the market.
6. Post-Marketing Surveillance: After a drug is approved and launched, post-marketing surveillance is conducted to monitor its long-term safety and effectiveness in real-world settings. This phase involves pharmacovigilance activities, such as monitoring adverse events, evaluating drug interactions, and assessing any additional benefits or risks associated with the drug.
It’s important to note that these phases may vary in duration and complexity depending on the specific drug, therapeutic area, and regulatory requirements. Additionally, some companies may engage in additional stages, such as reimbursement negotiations and ongoing research for new indications or formulations.